
Application Note:
Combustion-Based Radical Oxidation

Oxidation of silicon surfaces is a well-studied phenomenon with tremendous benefits in many aspects of silicon technology. The mainstay of oxidation processes in microelectronic applications for decades has been the use of so-called dry and wet thermal oxidation, in which silicon surfaces (typically planar wafer surfaces) are oxidized by O2- or H2O-containing ambient gases surrounding the wafers at temperatures often in excess of 1000 deg. C. Recently various methods of radical-based silicon oxidation have been developed to allow oxidation at lower temperatures and to improve uniformity of oxidation rates on different silicon crystal surfaces (e.g. <100> and <111>). Oxygen or hydroxyl radicals have been shown to oxidize silicon at lower temperatures and more evenly on all silicon surfaces than either O2 or H2O.
Among the various methods for generating oxidizing radicals (including plasma sources and so-called in-situ steam generation, or ISSG) one of the most attractive is radical generation via hydrogen combustion. In this method, the short-lived oxidizing radicals O and OH are created as intermediates in the overall reaction 2H2 + O2 = 2H2O. The reactants are inexpensive, and if the oxidation can be performed in a hot-wall furnace the hardware costs are not prohibitive as well. Proper selection of gas flow rates, operating pressures and temperatures can produce a regime in which both the spatial distribution of these intermediates and their volume density are adequate for oxidizing silicon. These are critical determinations, because the wafers must be exposed to the flowing gas stream at precisely the right moment—too early and the intermediates have not yet been generated; too late and the wafers see only H2O and wet oxidation results. Optimizing the process conditions and the wafer processing hardware can be daunting challenges, especially given the explosive nature of the reactants.
nVision Engineering have experience in both hardware and process development for combustion-based radical oxidation. Simulation-based design and appropriate simulation tools are the keys. While a complete reaction mechanism is much more complex than 2H2 + O2 = 2H2O, an adequate combustion mechanism can be assembled from the hydrocarbon combustion data from research supported by the Gas Research Institute (http://www.me.berkeley.edu/gri-mech/).
This mechanism can be employed to explore the process space and determine pressure and temperature ranges and gas flow rates that maximize the concentration and duration of the oxidizing radical species.
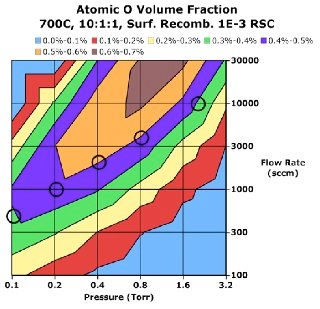
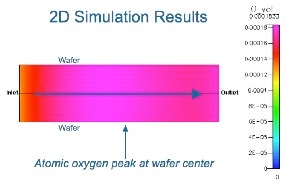
Having identified a process window, one can begin to optimize the hardware to achieve good results. For example, axial gas flow patterns employed in most batch tube furnaces are susceptible to depletion of radical species as described above. Hardware can be designed to minimize this depletion and yield improved on-wafer results. Most notably, Aviza Technology’s RVP-550™ radical oxidation systems (designed by nVision engineers) employ a cross-wafer flow path to avoid axial depletion effects and yield outstanding wafer-to-wafer uniformity. These reactors employ critical features in the reactor quartzware to assure uniform exhaust as well as optimized linear injector metering tubes to assure good injection flow uniformity for the desired process point (described in a separate Application Note). Both were designed using CFD as the primary design driver.
See also:
-
•T. Qiu, C. Porter, M. Mogaard, J. Bailey, H. Chatham. “Batch furnace radical oxidation,” Solid State Technology, October 2006 (Download PDF)
-
•J. Bailey, T. Qiu, H. Chatham, H. Treichel, and K. Mohamed. “Derivation of Rate Constants for the Batch Furnace Radical Oxidation of Silicon Wafers via Hydrogen Combustion,” J. Electrochem. Soc., 156, H372- H377 (2009) (Download PDF)
All Content Copyright 2011 nVision Engineering LLC. All rights reserved.